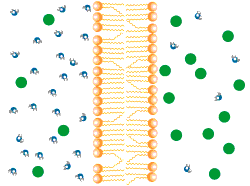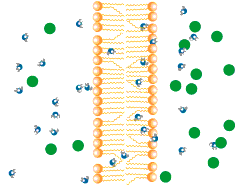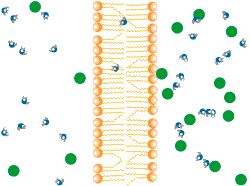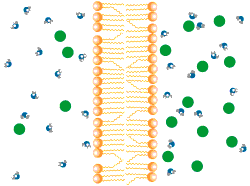
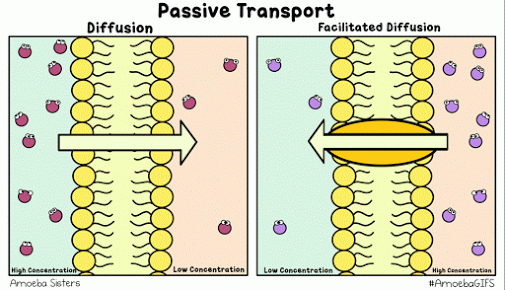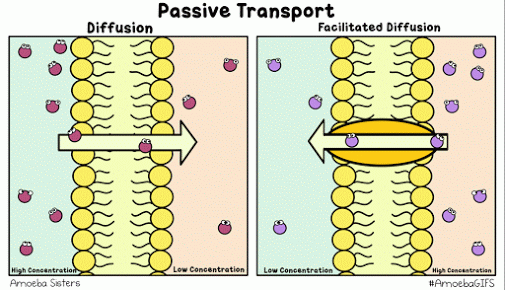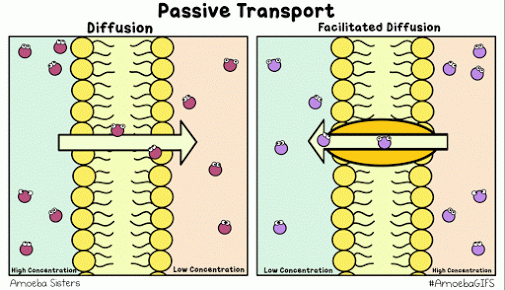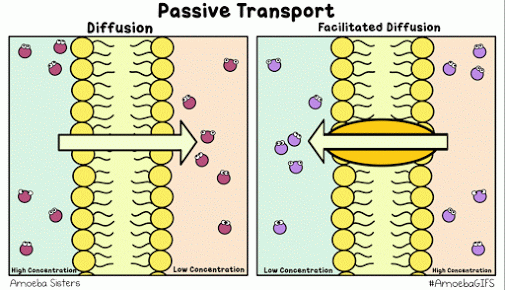
Diffusion describes the tendency of materials (solutes in particular) to want to move from areas of high concentration to areas of low concentration.
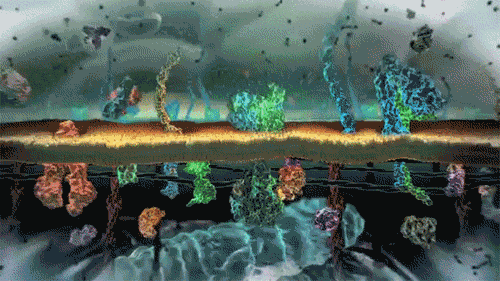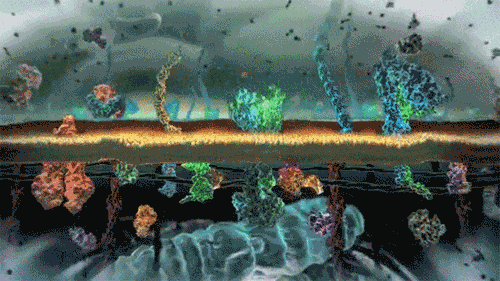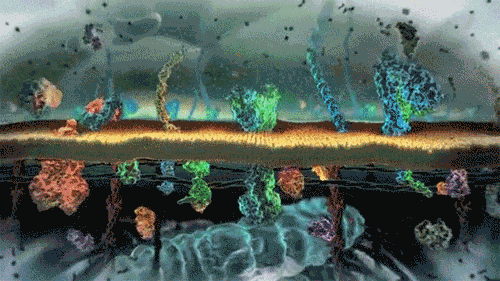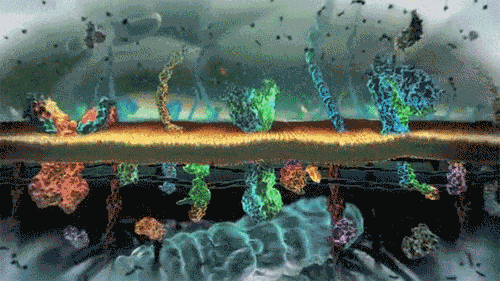
In the world of cells, often what is separating different solutions is the cell membrane made of a lipid bilayer and various different proteins.
The cell membrane is selectively permeable, only allowing certain molecules through. Usually, large and charged molecules have a difficult time crossing the lipid bilayer. Regardless, many of those solutes may still be attempting to cross the membrane so as to find areas of low concentration, ultimately striving for equilibrium (balanced concentrations)
The diffusion of water (or osmosis) is special in that water can cross most membrane, but many of its solutes (sugar for example) cannot. Water is often seeking low water concentration/high solute concentration areas to diffuse into. Generally, the cell membrane allows for the passage of water.
Many proteins act as channels to allow specific molecules through. In facilitated diffusion, a protein channel allows a specific molecule to diffuse across the membrane from a high to low concentration. This requires no energy and is often the only way certain molecules can cross the membrane (again, due to their size or charge).
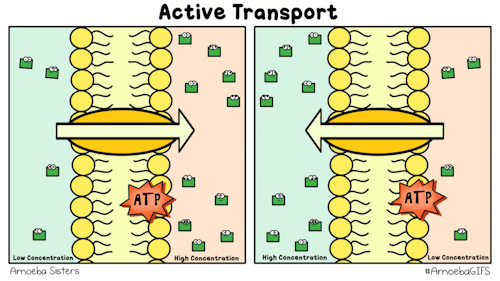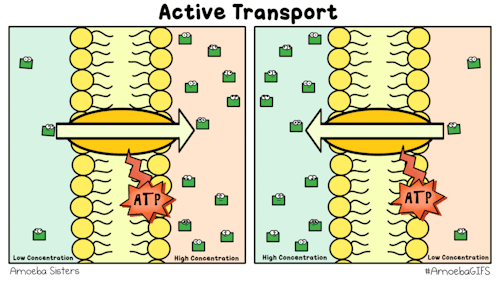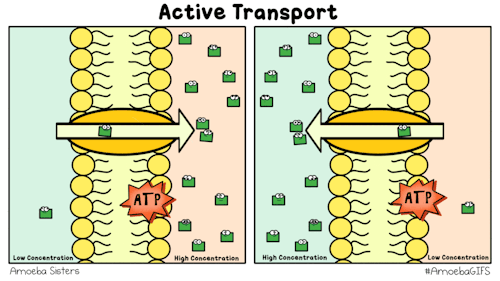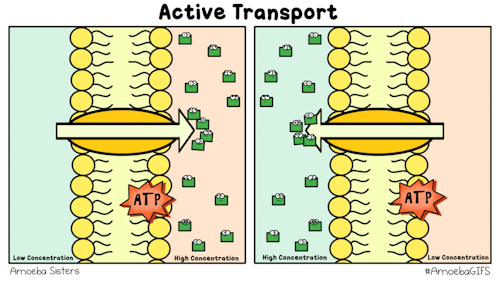
Yet, other protein channels perform active transport where they move specific molecules across the membrane from a low concentration to a high concentration. Because this is against the natural flow of solutes, this requires the cell to use energy.














 RSS Feed
RSS Feed